A municipality recently discovered that one of its wells was contaminated with the compounds listed in the following:
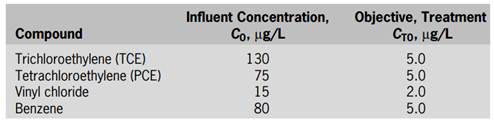
To continue using the well as a drinking water resource, the compounds shown in the above table need to be removed to meet the treatment objectives shown in the table. During normal pumping operations, the well produces about 2.18 ML/d, and further expansion of the well field may be considered depending on the efficacy of the ozone/hydrogen peroxide process. The pH, and NOM concentrations are 7.5, 400 mg/L as CaCO3, and 1.2 mg/L as C, respectively (at this pH and alkalinity, HCO3 − = 487 mg/L and CO3 2− = 0.71 mg/L). Important physicochemical properties for the compounds that need to be removed are as follows:
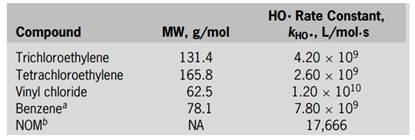
a Molar extinction coefficient is high but quantum yield is very low; consequently, photolysis can be ignored. bFor NOM, the unit of kHO• is L/mg·s.
For simplicity, a proprietary reactor will be used. Based on dye studies, it has been found that the reactor can be modeled as four completely mixed reactors in series. The reactor is 1 m in diameter and 3 m in height, and the volume is approximately 2300 L. For the given conditions, determine the optimum H2O2 dosage to achieve the treatment objectives based on the simplified model (Sim-PSS). Consider ozone dosages of 1, 3, and 5 mg/L.
Students succeed in their courses by connecting and communicating with an expert until they receive help on their questions

Consult our trusted tutors.